DENMARK
Danish BioImaging
Danish BioImaging (DBI) is a multi-sited, multimodal Node bringing together five facilities across the country, representing the joined national imaging infrastructure of Denmark. The Node provides a broad service offer covering a wide range of advanced and state-of-the-art bioimaging technologies - from pre-clinical imaging of big animals and humans to cryo-electron microscopy for single particle analysis- and fields of expertise – covering plant biology, HCS of yeast libraries, zoology, pathology, neurosciences and metabolism. The increase in size and complexity of image-based data sets produced by single and multimodal bioimaging technologies makes image analysis a key part of the DBI consortium. To support data storage, management and image analysis needs, DBI includes a national Image analysis service.
Specialties and expertise of the Node
- Dedicated MRI, CT, ultrasound and PET systems at the world’s largest experimental pig surgery facility
- Exotic animal models in vivo and ex vivo pre-clinical imaging
- A unique animal imaging data repository, covering preclinical imaging data from more than 5000 species.
- Metabolism imaging in whole animals using hyperpolarized MRI and PET (Incl. specialized radiochemistry and novel PET tracers laboratory)
- Imaging cardiac and renal oxygen metabolism
- Cardiovascular live imaging (4D MRI and 4D ultrasound imaging of flow patterns, CT imaging of cardiac electric conduction networks and imaging myocardial blood perfusion with PET)
- Bioimaging models and applications in Neuroscience
- 3D printing of organs, animals and personalized human skin (Incl. skin cancer models)
- Blood barrier models based on primary porcine cells and/or human stem cells
- Plant live imaging (including plant growth visualization) and hystology
- Light Microscopy super resolution expertise and dedicated microscopes to perform MP- STED, FCS-STED, FLIM-STED and stimulated Raman scattering microscopy
- Molecule dynamics analysis with CARS and spatial transcriptomics using MERFISH to simultaneously measure the amount and spatial distribution of hundreds to thousands of RNA species in single cells
- Screening of large genome-wide yeast libraries
- Deep imaging of organs and organisms with SPIM
- Correlative Light and Electron Microscope pipelines
- Image-based Computer science expertise and computing infrastuctures
 Green mitochondria on fat: The inside of a brown fat cell with multilocular lipid droplets (white and brown) surrounded by mitochondria and other smaller organelles (green). Dual beam SEM with FEI Quanta FEG 3D and 3D rendering with Amira. Elahu Gosney Sustarsic
Assistant Professor, Novo Nordisk Foundation Center for Basic Metabolic Research,
University of Copenhagen
Green mitochondria on fat: The inside of a brown fat cell with multilocular lipid droplets (white and brown) surrounded by mitochondria and other smaller organelles (green). Dual beam SEM with FEI Quanta FEG 3D and 3D rendering with Amira. Elahu Gosney Sustarsic
Assistant Professor, Novo Nordisk Foundation Center for Basic Metabolic Research,
University of Copenhagen
Offered Technologies
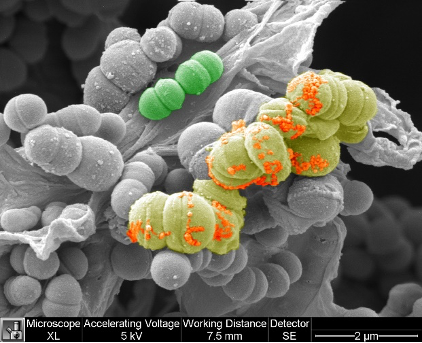 Streptococcus imaged with SEM FEI XL30 and pseudocolored with photoshop by Professor Mattias Mörgelin, University of Lund
Streptococcus imaged with SEM FEI XL30 and pseudocolored with photoshop by Professor Mattias Mörgelin, University of Lund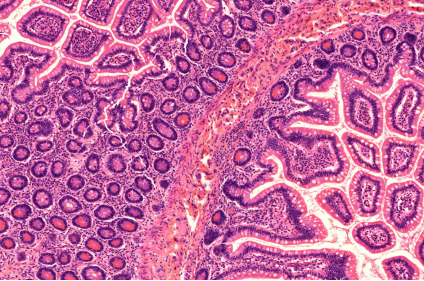 Human intestine section stained with Hematoxylin-Eosin and imaged with a Zeiss Axioscan Z1. Histology Laboratory at the Core Facility for Integrated Microscopy
Human intestine section stained with Hematoxylin-Eosin and imaged with a Zeiss Axioscan Z1. Histology Laboratory at the Core Facility for Integrated MicroscopyList of offered technologies:
| Technologies | Euro-BioImaging |
| Deconvolution widefield microscopy (DWM) | ✓ |
| Laser scanning confocal microscopy (LSCM/CLSM) | ✓ |
| Spinning disk confocal microscopy (SDCM) | ✓ |
| Structured illumination microscopy (SIM)* | ✓ |
| Total internal reflection fluorescence microscopy (TIRF) | ✓ |
| Two-photon microscopy (2P) | ✓ |
| Image Scanning microscopy (ISM) | ✓ |
| cryoFM * | ✓ |
| Single Molecula localisation microscopy (SMLM) | ✓ |
| Stimulated emission depletion microscopy (STED) | ✓ |
| Light-sheet mesoscopic imaging (SPIM or dSLSM) | ✓ |
| Coherent Anti-Stokes Raman Scattering microscopy* (CARS) | ✓ |
| Raman Spectroscopy (RS) | ✓ |
| Fluorescence (cross)-correlation spectroscopy (FCS/FCCS) | ✓ |
| Fluorescence Resonance Energy Transfer (FRET) | ✓ |
| Fluorescence Recovery After Photobleaching (FRAP) | ✓ |
| Fluorescence Lifetime Imaging Microscopy (FLIM) | ✓ |
| TEM of chemical fixed samples (TEM) | ✓ |
| TEM of cryo-immobilized samples (TEM cryo samples)* | ✓ |
| EM tomography (ET) | ✓ |
| FIB-SEM | ✓ |
| Serial Blockface SEM | ✓ |
| Immuno-gold EM on thawed cryo-sections (Tokuyasu-EM) | ✓ |
| Immuno-gold EM on resin sections (resin-EM) | ✓ |
| Cryo Transmission Electron Microscopy (Cryo-TEM)* | ✓ |
| Scanning Electron Microscopy (SEM) | ✓ |
| micro-MRI/MRS (>= 7T) | ✓ |
| micro-MRI/MRS (< 7T) | ✓ |
| micro-CT | ✓ |
| micro-PET | ✓ |
| micro-SPECT | ✓ |
| micro-US | ✓ |
| in vivo Optical Imaging | ✓ |
| micro-PET/MRI | ✓ |
| micro-MRI/MRS (>= 7T) - ex-vivo | ✓ |
| micro-MRI/MRS (< 7T) - ex-vivo | ✓ |
| micro-CT - ex-vivo | ✓ |
| Image Analysis-bio * | ✓ |
| Image Analysis-med * | ✓ |
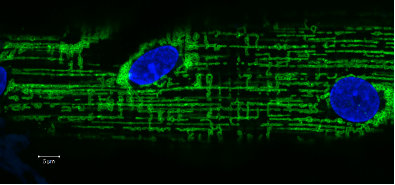 Human vastus lateralis single muscle fiber stained against mitochondrial networks (Green) and myonuclei (blue). Images acquired with a Zeiss LSM700 by Associate Professor Clara Prats, University of Copenhagen
Human vastus lateralis single muscle fiber stained against mitochondrial networks (Green) and myonuclei (blue). Images acquired with a Zeiss LSM700 by Associate Professor Clara Prats, University of Copenhagen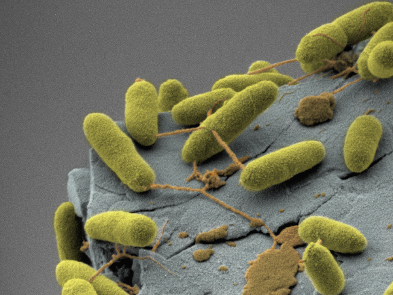 Bacteria imaged in a FEI Quanta 3D FEG and pseudocolored with Amira software
Bacteria imaged in a FEI Quanta 3D FEG and pseudocolored with Amira software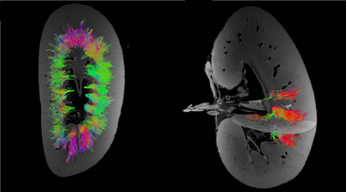 MRI of pig kidneys, visualization of tubule structures. Method: MRI clinical system and diffusion tensor sequence, Aarhus University Hospital
MRI of pig kidneys, visualization of tubule structures. Method: MRI clinical system and diffusion tensor sequence, Aarhus University Hospital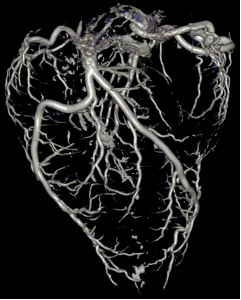 CT of giraffe heart, visualization of blood vessels. Method: CT clinical system and iodine filling, Aarhus University Hospital
CT of giraffe heart, visualization of blood vessels. Method: CT clinical system and iodine filling, Aarhus University Hospital OCT of mouse brain cortex, visualization of cerebral vessels. Method: Preclinical Fourier-domain OCT imaging with en face maximum intensity projection, Aarhus University Hospital.
OCT of mouse brain cortex, visualization of cerebral vessels. Method: Preclinical Fourier-domain OCT imaging with en face maximum intensity projection, Aarhus University Hospital.Additional services offered by the Node
- Project planning and methodological setup
- Wet labs
- Cell culture facilities
- Animal housing
- Imaging probes
- Data processing and analysis
- Data storage
Contact details
Clara Prats cprats@sund.ku.dk
Sonia Diaz Garcia sonia.garcia@sund.ku.dk
www.danishbioimaging.dk