March 26, 2026
Welcome, MAdMiC Node!
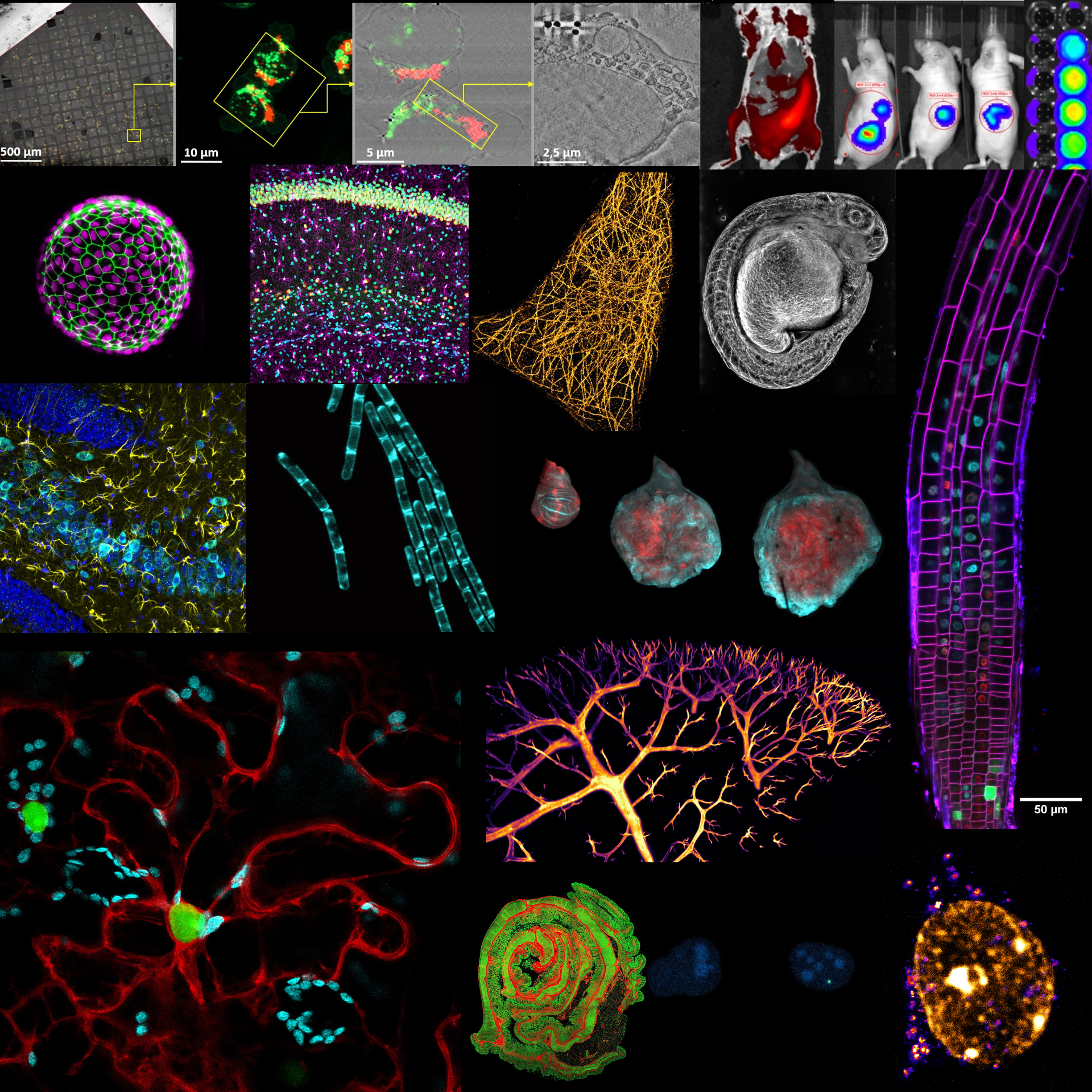
The Madrid Advanced Microscopy Center (MAdMiC) is the first Euro-BioImaging Node in Madrid (Spain). It is formed through the collaboration and close work of…
Clinical evaluation of novel treatments and comparisons with existing ones is of paramount importance in the development of drugs and therapies, to ensure their efficacy and safety. In most cases, Phase III clinical trials are conducted simultaneously by more than one independent medical institution, allowing the enrolment of a larger number of participants at a faster rate and increasing the heterogeneity of the population cohorts.
The Brain Imaging Network (BIN) Node in Portugal is very active in this field and recently it received a number of requests for access from different companies that are involving medical centers from around the globe in clinical trials for testing their products.
Examples include multicenter clinical trials to evaluate the efficacy and safety of treatments of Alzheimer’s and Parkinson’s diseases and different types of tumors, that were commissioned, among other companies, by Biogen, GemVax & Kael, Bial, Gilead, Olema Pharmaceuticals, GlaxoSmithKline, Bayer, Merck Sharp & Dohme Lda (https://clinicaltrials.gov/).
These companies chose the BIN Node as it is fully equipped with relevant PET and MRI instrumentation and its staff is very experienced both in the imaging part and in the related issues such as subjects recruitment, ethics and legal management, image data processing and analysis. Its specific skills in the neuroscience and oncology fields and the strong linkages with the the local health services providers and with the Institute of Nuclear Sciences Applied to Health (ICNAS) at the University of Coimbra make the BIN Node an ideal candidate to support clinical trials where PET and MR Imaging is needed.
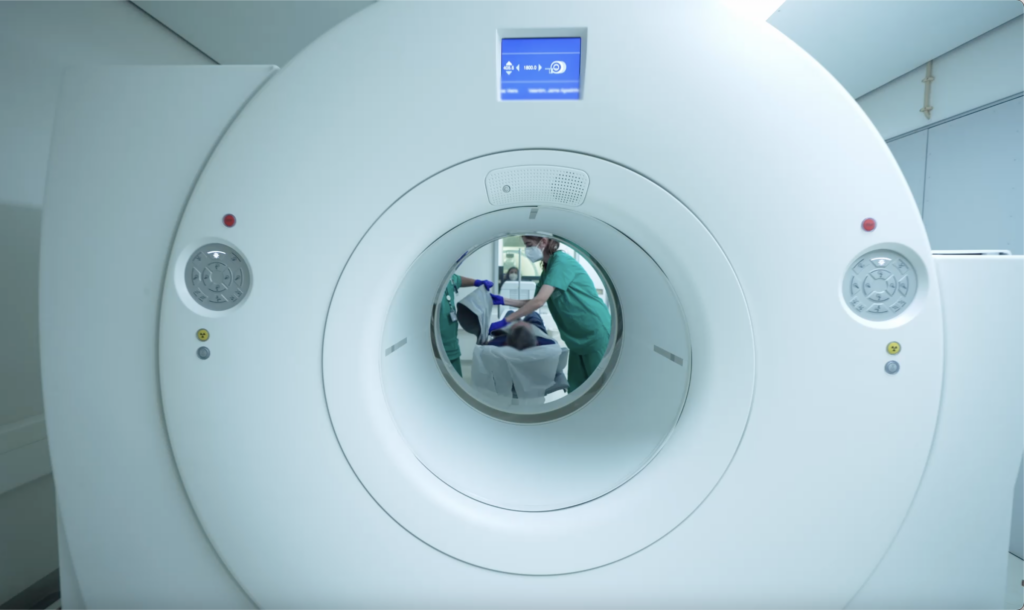
Prof. Miguel Castelo Branco, the Head of the BIN Node, explains: "Taking part in clinical trials requires an ability to coordinate across sites respecting the deadlines. At the Portuguese BIN Node we prioritise this type of collaboration and ensure the confidentiality of our user projects. If you are working on neuroscience, cardiology or oncology applications, we welcome you to get in touch."
Article written by Alessandra Viale
Learn more about the expertise and technologies available at the Portuguese BIN Node (video in Portuguese).

March 26, 2026
The Madrid Advanced Microscopy Center (MAdMiC) is the first Euro-BioImaging Node in Madrid (Spain). It is formed through the collaboration and close work of…

March 26, 2026
German BioImaging, within its work in the NFDI4BIOIMAGE consortium, and in collaboration with Euro-BioImaging ERIC, has launched a new survey to collect input about…

March 25, 2026
Turin, Italy – 20–22 October 2026 Early career professionals working in imaging core facilities will soon have the opportunity to strengthen essential skills beyond…