
May 22, 2026
Austrian BioImaging celebrates its 11th anniversary
On 11 May 2026, Austrian BioImaging/CMI celebrated its 11th anniversary with a special event at the SkyLounge of the University of Vienna, bringing…
Francois Lux is an Associate Professor of Chemistry at the University of Lyon 1. His research interests include nanomedicines – precisely the development of nano drugs for imaging therapy and theranostics and biopolymers. Imaging, and in particular MRI, is a fundamental part of his research. In a recent project, he worked with the Molecular Imaging Italian Node to study the biodistribution, tumor uptake and excretion of a special polymer he and his colleagues developed for drug delivery in anticipation of clinical applications. Satisfied with the results, he started a second project with our Node to explore the potential of the polymer for targeted therapy action. Learn more about this exciting collaboration in the interview below.
Francois Lux and his group develop hydrogels that can be used for drug delivery and captation of metals. Typically, these gels are developed in the laboratory and tested on mice. While the ultimate goal is to use these in deliverance of immunotherapies to treat human illness like cancer, they must pass many tests before being used in the clinic. “When it comes to human health, excretion is key,” explains Francois Lux. “For some hydrogels, the requirement is that they should be eliminated in one month. So we develop our materials and test different formulations on mice to see if some residue is still in the body at different time points.”
Most recently, Francois and his group have been working on a polymer made of partially acetylated chitosan and functionalized with DOTAGA, a chelate which is used as MRI positive contrast agent when combined with gadolinium. This polymer has high potential for drug delivery. But before it can be translated to the clinic, it must be confirmed that the polymer is not toxic, biodegradable and undergoes rapid renal excretion.
The Molecular Imaging Center (CIM) at the University of Torino, part of Euro-BioImaging’s MMMI Italian Node, is specialized in experiments that combine chemistry with imaging. They provide turn-key project solutions that range from synthesizing compounds to preparing animal models to data analysis. Exactly the kind of partner Francois was looking for.
“I had met Enzo Terreno and other colleagues from the CIM facility at the University of Torino multiple times at conferences, but we never had the opportunity to work together,” says Francois. “When Enzo told me about the Italian User Access Fund, I decided to apply. This funding provided us with a catalyst for collaboration.”
Like many preclinical imaging projects, Francois Lux’s project was completely remote. Enzo Terreno and his group have a tried and true protocol for working remotely to provide preclinical imaging services (read the article here).
“We had lots of exchanges before sending in the proposal, we had already determined the project protocol before I applied for funding,” explains Francois. “And during the actual project lifetime, we were able to work together by email and zoom.”
While Francois followed the advancements from Lyon, a multidisciplinary staff with expertise ranging from chemistry, to biology of cells to animal handling and surgery, carried out the experiments in Torino.
They used MRI to look at the amount of gel in the animals, measuring the volume of the gel over time.
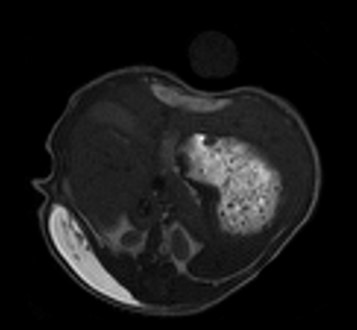
“MRI is a very good technology to visualize the decrease of the size of the gel over time after administration in mice,” states Francois. “It is very interesting to follow preclinically what can be the kinetics of the formulation. In addition, MRI images provide information about distribution of the polymer in the different organs and tumor uptake.”
Francois Lux is happy with the results from his first project. “There’s a good twinning of the hydrogel. All data indicate that the polymer is biodegradable and is eliminated within one month, as shown by MRI and confirmed by optical imaging and ICP-MS. But in our first experiment, the hydrogel was empty (with no antibody) and no histology was done at the end of the protocol to see if the degradation was complete and to assess the inflammation status. So, we undertook a second project with the University of Torino to complete our findings,” he explains.
The results of these projects are very promising and Francois is now planning to apply for a third access at the Node to study new formulations of the polymer.
While we wait to find out if this new polymer will pass the requirements for use in clinical applications, we are happy that a new collaboration has been born, thanks to the Euro-BioImaging Italian User Access Fund.

May 22, 2026
On 11 May 2026, Austrian BioImaging/CMI celebrated its 11th anniversary with a special event at the SkyLounge of the University of Vienna, bringing…

May 22, 2026
The 14th Euro-BioImaging Board meeting took place in Prague on 20–21 May 2026, hosted by Czech BioImaging. The meeting provided an opportunity for…
May 21, 2026
Understanding the effects of ionizing radiation on developing organisms is a critical area of research in radiobiology and developmental biology. Embryonic tissues are…