March 26, 2026
Welcome, MAdMiC Node!
The Madrid Advanced Microscopy Center (MAdMiC) is the first Euro-BioImaging Node in Madrid (Spain). It is formed through the collaboration and close work of…
Imaging technologies can help scientists understand how plant tissues respond to stress, such as drought, heat, or other environmental factors. Characterising the mechanical properties of plant cells is an important aspect of studying plant resilience. That is why a team at Wageningen University, host of Euro-BioImaging’s Advanced Light Microscopy and Molecular Imaging Node Wageningen, developed a new method to measure the viscosity in individual parts of plant cells using Fluorescence Lifetime Imaging Microscopy (FLIM). In this article, Professors Dolf Weijers and Joris Sprakel of Wageningen University describe their method and discuss its implications.
There is an increasing body of evidence that mechanical signals, e.g. those exerted by the growth and division of cells on their neighbors, act as instructive biological signals that trigger biochemical processes. For example, the orientations of cell division, and thereby tissue geometry, are now known to be controlled in part by mechanical interactions between adjacent cells. Understanding how the laws of mechanics shape tissues and instruct a wide diversity of biological processes is one of the central aims in the emerging field of plant mechanobiology. However, it remained unclear how these mechanical patterns could be directly, and quantitatively measured. This was the problem we addressed in this project: How can we map mechanical patterns in plant cells and tissues quantitatively, in-situ and with high resolution?
To resolve this methodological challenge, we combined FLIM imaging with a new class of fluorescent molecular sensors we developed, whose optical signature changes in response to changes in the viscosity of the material surrounding the probe. The molecules where designed to locate themselves to specific parts of the plant cell, which allowed us to perform local mechanical measurements in these areas. The use of these molecular reporters, in conjunction with quantitative FLIM, enabled us to construct the first microviscosity maps of plant cells and tissues.
These probes were specifically designed to work with FLIM, as this technique offers the possibility of doing highly quantitative measurements of the optical properties of a probe, much more so than ratiometric imaging approaches, while still offering the imaging advantages of a confocal microscope. FLIM is simply the best technique to leverage an accurate measurement with a high spatial resolving power.
A complete description of our approach can be found in the article, “Complete microviscosity maps of living plant cells and tissues with a toolbox of targeting mechanoprobes,” published in PNAS.
Our future work will be directed at employing the technique to answer new biological questions, such as what role of mechanical signalling plays in plant embryogenesis and what mechanisms plant cells utilize to accommodate external mechanical stimulation. Moreover, we are exploring the use of this approach also in other organisms, such as plant pathogens that exploit mechanical stress to gain entry into their host and in which the visualization of the phenomena could bring new light to how pathogenic invasion is achieved.
Recently our Wageningen UR Microspectroscopy Centre (part of the Wageningen node) invested in a new instrument capable of rapid multiphoton 3D FLIM imaging, which opens the opportunity to look deeper inside opaque tissues and monitor faster processes. Multiphoton imaging will allow us to explore and visualize biology in regions that have remained hidden so far.
Euro-BioImaging is the European landmark research infrastructure for biological and biomedical imaging as recognised by the European Strategy Forum on Research Infrastructures (ESFRI). All scientists, regardless of their affiliation, area of expertise or field of activity can benefit from Euro-BioImaging’s pan-European open access services. By facilitating user access to high quality imaging facilities, resources, and services, with a constantly evolving technology offer, Euro-BioImaging will boost the productivity and impact of research across Europe.
Potential users of the Euro-BioImaging technologies are encouraged to submit project proposals via our website. To do so, you can log in to access our application platform, choose the technology you want to use and the facility you wish to visit, then submit your proposal. By using Euro-BioImaging, users will benefit from advice and guidance by technical experts working at the Nodes, training opportunities, and data management services.
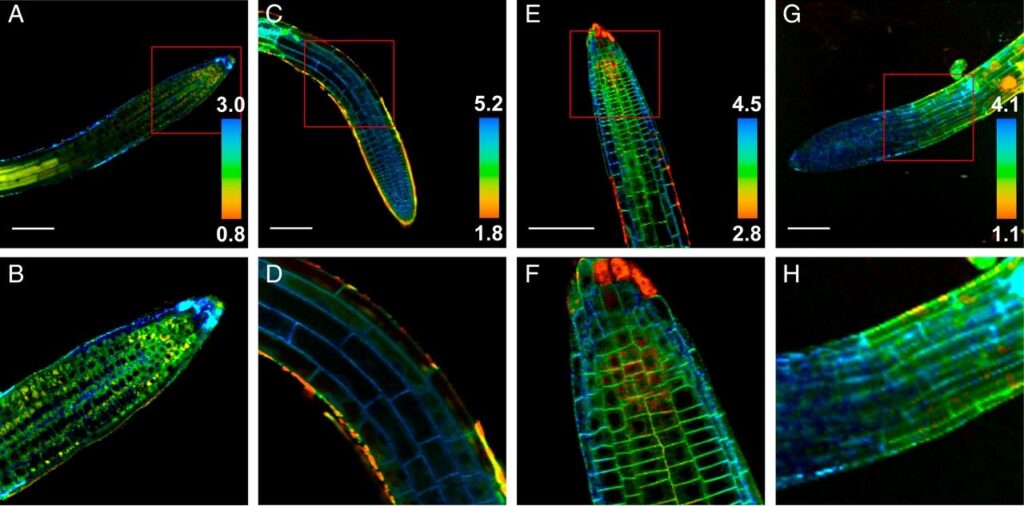
Figure: Fluorescence lifetime microviscosity maps of Arabidopsis roots showing the vacuoles (sulfo-BDP, A and B), cytosol (PEG-BDP, C and D; note that the dye penetrates poorly in the meristem region), plasma membranes (N+-BDP, E and F), and cell walls (CWP-BDP, G and H), in all images. (Scale bar: 100 μm.) Red boxes in A, C, E, and G indicate close-ups shown in the B, D, F, and H, respectively. https://creativecommons.org/licenses/by-nc-nd/4.0/legalcode
Citation:
Complete microviscosity maps of living plant cells and tissues with a toolbox of targeting mechanoprobes. Lucile Michels, Vera Gorelova, Yosapol Harnvanichvech, Jan Willem Borst, Bauke Albada, Dolf Weijers, Joris Sprakel. Proceedings of the National Academy of Sciences, Jul 2020, 117 (30) 18110-18118; DOI: 10.1073/pnas.1921374117
Learn more:
https://www.wur.nl/en/show/Breakthrough-in-research-into-mechanical-forces-in-plant-cells.htm
https://www.pnas.org/content/117/30/18110

March 26, 2026
The Madrid Advanced Microscopy Center (MAdMiC) is the first Euro-BioImaging Node in Madrid (Spain). It is formed through the collaboration and close work of…

March 26, 2026
German BioImaging, within its work in the NFDI4BIOIMAGE consortium, and in collaboration with Euro-BioImaging ERIC, has launched a new survey to collect input about…

March 25, 2026
Turin, Italy – 20–22 October 2026 Early career professionals working in imaging core facilities will soon have the opportunity to strengthen essential skills beyond…