
April 13, 2026
We are hiring a Data Architect/Knowledge Engineer
Euro-BioImaging Bio-Hub team, hosted by EMBL in Heidelberg, is hiring a Data Architect/Knowledge Engineer – Euro-BioImaging (AI4Access) to…
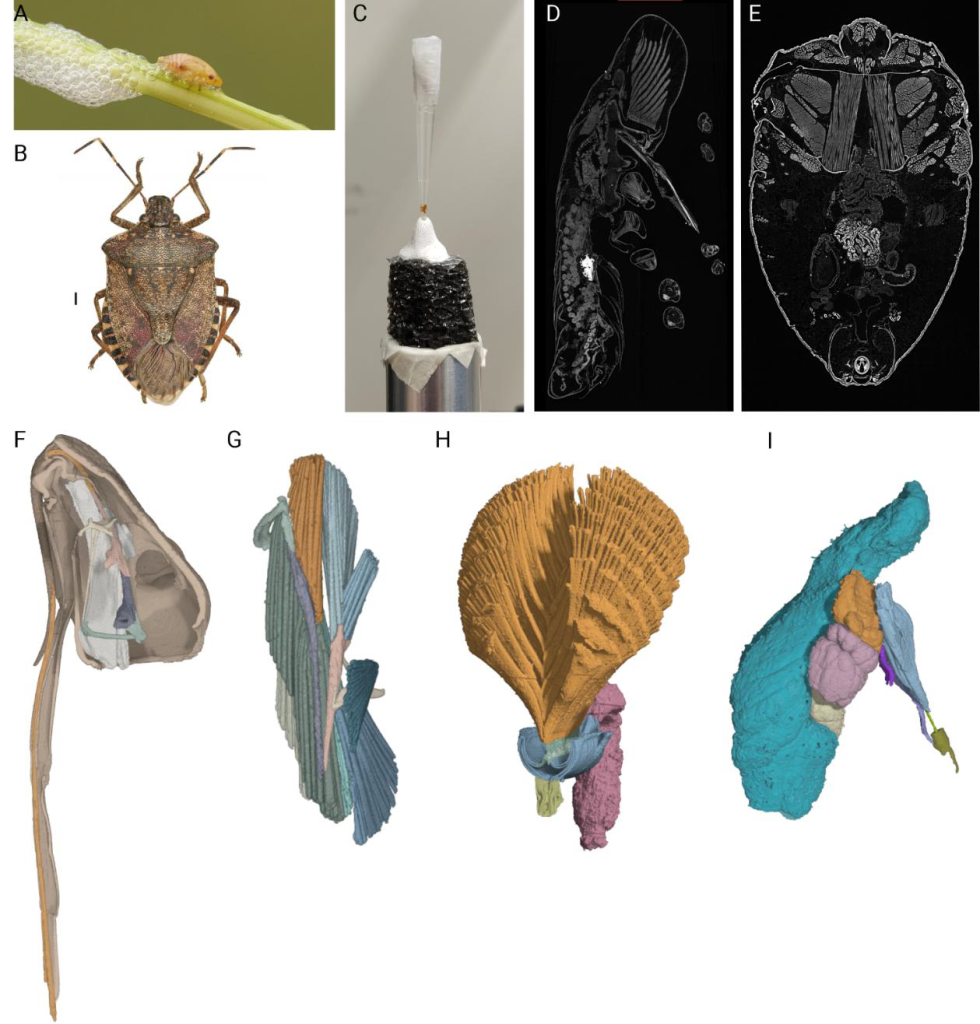
The Olive Quick Decline Syndrome is a complex disease inducing leaf scorch and dieback symptoms on olives, caused by the xylem-limited bacterium Xylella fastidiosa subsp. pauca ST53. In Italy, the bacterium has arrived in the Apulia region, infecting a major part of it. The bacterium proliferation in southern Italy is partially facilitated by the presence of insects, which are the vectors of Xylella fastidiosa. Until now, Philaenus spumarius, the meadow spittlebug, has been recognized as the primary vector of Xylella fastidiosa. A unique characteristic of this insect is foam production when at the nymphal stage. The foam has a protective role for the immatures and can serve as a medium for intraspecific communication. Therefore, the elucidation of the mechanisms of foam production and release and a detailed knowledge of the morphology and physiology of the reproductive system in this insect is very important, as it may help establish control measures against the propagation of Xylella fastidiosa. Nevertheless, only a few studies have been made years ago, and relevant information is still lacking.
Roberto Romani is associate professor at the Department of Agricultural, Food and Environmental Sciences at the University of Perugia. Milos Sevarika is a postdoctoral researcher in the same department. Their research work focuses on the physiology, functional anatomy and behavior of insects. In order to explore the mechanisms of foam production and the morphology and physiology of the reproductive system in Philaenus spumarius. Together, they requested access to the Phase Contrast Imaging Node in Trieste.
A primary challenge in the study of insect structural organization is the inherently destructive nature of traditional anatomical methods. Historically, investigating internal morphology has relied on micro-dissection and histological serial sectioning. Although these techniques provide good resolution, they require the physical destruction of the specimen, making it impossible to visualize complex spatial representations.

Non-destructive X-ray methods offer a solution to these limitations; however, conventional X-ray imaging often yields unsatisfactory results for internal soft tissues (e.g., the brain, digestive tract, and muscles), given that biological organs have similar x-ray attenuation coefficients, which makes them appear as featureless and translucent. This lack of contrast becomes even more prominent in larger, whole-body, or heavily sclerotized specimens, where the dense cuticle further obscures the subtle density variations of the internal organs.
To preserve structural organization while increasing tissue contrast, one can apply chemical fixatives traditionally used for electron microscopy coupled with Lugol’s solution. This combination ensures that delicate internal features are fixed while providing the contrast necessary to distinguish between various soft-tissue types. Furthermore, as the cuticle can act as a significant barrier and source of artifacts in standard imaging, the application of Phase-Contrast X-ray Tomography can significantly enhance the intrinsic contrast between organs. Propagation based Micro-CT imaging enhances the phase shift of the X-ray beam as it passes through different refractive indices, allowing for the visualization of fine morphological details that remain invisible under conventional X-ray settings.
By accessing the Phase Contrast Imaging Node Elettra in Trieste, Professor Romani and Milos Sevarika were able to analyse Philaenus spumarius in various developmental stages, using Lugol’s iodine solution staining and Phase-Contrast X-ray Tomography at our Node, with three big advantages.
First, the flexible set up available at the SYRMEP beamline was very useful, as the samples often exceeded the vertical field of view and had to be mounted on a vertically controlled holder and scanned with a partially overlapping scanning mode.
Second, the use of a prolonged Lugol’s solution based staining procedure ensured that internal tissues and muscles absorbed sufficient iodine, resulting in higher X-ray attenuation and a clear delineation between structures. And third:
The sub-micron resolution (0.65 µm) and phase-contrast enhancement at the SYRMEP beamline allowed for a detailed characterization of the internal organization, which was essential for studying the complex nymphal anatomy of P. spumarius as well as the entire insects.
-- Roberto Romani, University of Perugia
The internal 3D investigation of immatures and adult individuals of P. spumarius provided new insights into the mechanism of foam production and reproductive system organization, development and physiological evolution, and will contribute to obtain a comprehensive picture of these relevant biological aspects.
The same protocol was applied also to other insect species with a different size, such as the brown marmorated stink bug, Halyomorpha halys, which is responsible for damage to crops.
We would like to thank EuroBioimaging for providing access to the Node. We also would like to thank Dr. Giuliana Tromba and Dr. Elena Longo (Elettra, Trieste) for their professional support and help in setting up the system and assisting throughout the project.
-- Roberto Romani, University of Perugia

April 13, 2026
Euro-BioImaging Bio-Hub team, hosted by EMBL in Heidelberg, is hiring a Data Architect/Knowledge Engineer – Euro-BioImaging (AI4Access) to…

April 13, 2026
As part of our ongoing series spotlighting the scientists working at imaging facilities across the Euro-BioImaging realm, we sat down with Przemek Krawczyk, a…

April 9, 2026
Euro-BioImaging ERIC is deeply grateful to announce that the European Union has entrusted our infrastructure with funding to shape the future of imaging and…